Lab News
See what we've
been up to in
The Shankland Lab!
Watch our video and learn about the unique and rigorous
Nephrology Fellowship Program at the University of Washington
“Loss of Epigenetic Information as a Cause of Mammalian Aging”
Published in Cell
“PD-1 signaling in aged kidneys and diseased podocytes”
Jeffrey W Pippin, Natalya Kaverina, Yuliang Wang, Diana G Eng, Yuting Zeng, Uyen Tran, Carol J Loretz, Anthony Chang, Shreeram Akilesh,
Chetan Poudel, Hannah S Perry, Christopher O'Connor, Joshua C Vaughan, Markus Bitzer, Oliver Wessely, Stuart J Shankland
The Shankland Lab's research is featured on the cover of
The Journal of Clinical Investigation
2022 Pre-Doctoral Fellowship Award
Congratulations Yuting Zeng!
Institution: University of Washington
Project Title: Podocyte Inflammation Accelerates Parietal Epithelial Cell Injury in the Aged Kidney
Yuting describes her research in one sentence: My research focuses on using novel open microfluidic coculture platforms to investigate the intercellular crosstalk between injured podocytes and parietal epithelial cells (PECs) in the events of aging and glomerulosclerosis.
Learn more about Yuting’s work: kidneycure.org
ASN Kidney TREKS Program
Tutored Research and Education for Kidney Scholars
Huge congratulations to our PhD Student, Yuting Zeng for her
acceptance to the 2022 course!
The TREKS program will be held in the Mount Desert Island Biological Laboratory (MDIBL) in Bar
Harbor, ME. ASN will cover room, board, and tuition for Yuting to attend, as well as $500 in travel
support. Yuting will also be invited to become a Kidney STARS member, with complimentary
registration to ASN Kidney Week in Orlando, FL, and $1,000 in travel support to attend the conference.
Great job Yuting!!
Congratulations Dr. Shankland!
Dr. Stuart Shankland, recipient of the 2021 Marilyn Farquhar Award for Podocyte Biology
NephCure Kidney International 13th Annual Podocyte Conference
University of Manchester
Read more about Dr. Shankland and the Marilyn Farquhar Award at nephrology.uw.edu
12th International Podocyte Conference
“Podocyte Replacement in Disease”
presented by Dr. Stuart Shankland, MD, MBA
ASN - Kidney Week - New Orleans
Congratulations!
Gary Liu has earned his PhD
Lab member, Andrea Largent
discusses her research at the
20th Annual Undergraduate Research Symposium
Shankland Lab Research featured on cover of Kidney International
Lam member, Dr. Mariya Sweetwyne's paper accepted to Kidney International
Mitochondrial-targeted peptide, SS-31, improves glomerular architecture in mice of advanced age
PMID: 28063595
New Funding for FSGS Research
FY 2015 Investigator-Initiated Research Award – Partnering PI Option
Select Presentations
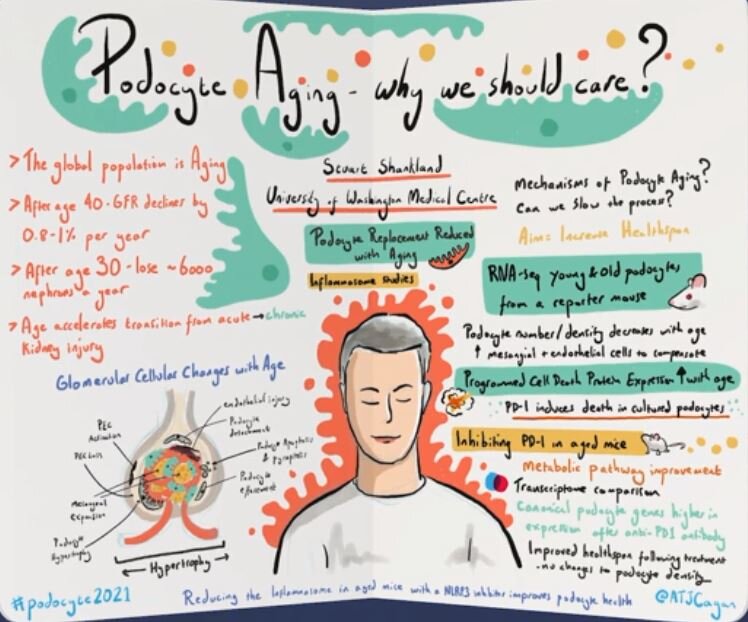
NephCure Kidney International Podocyte Conference - 2021
“Podocyte Aging - why we should care?”
Society of Toxicologic Pathology Annual Meeting - 2018
International Podocyte Conference - 2018
American Society of Nephrology Annual Meeting, New Orleans, LA - 2017
“Podocyte Regeneration from Renin Lineage Cells”
American Society of Nephrology Annual Meeting, San Diego, CA - 2015
"What is Aging Nephropathy?"
"Role of Parietal Cells Following Glomerular Injury"
New York Renal Society - 2015
German Society of Nephrology - 2014
International Podocyte Conference - 2014
National Kidney Foundation Annual Meeting - 2014
SSCI Annual Meeting - 2014
Posters
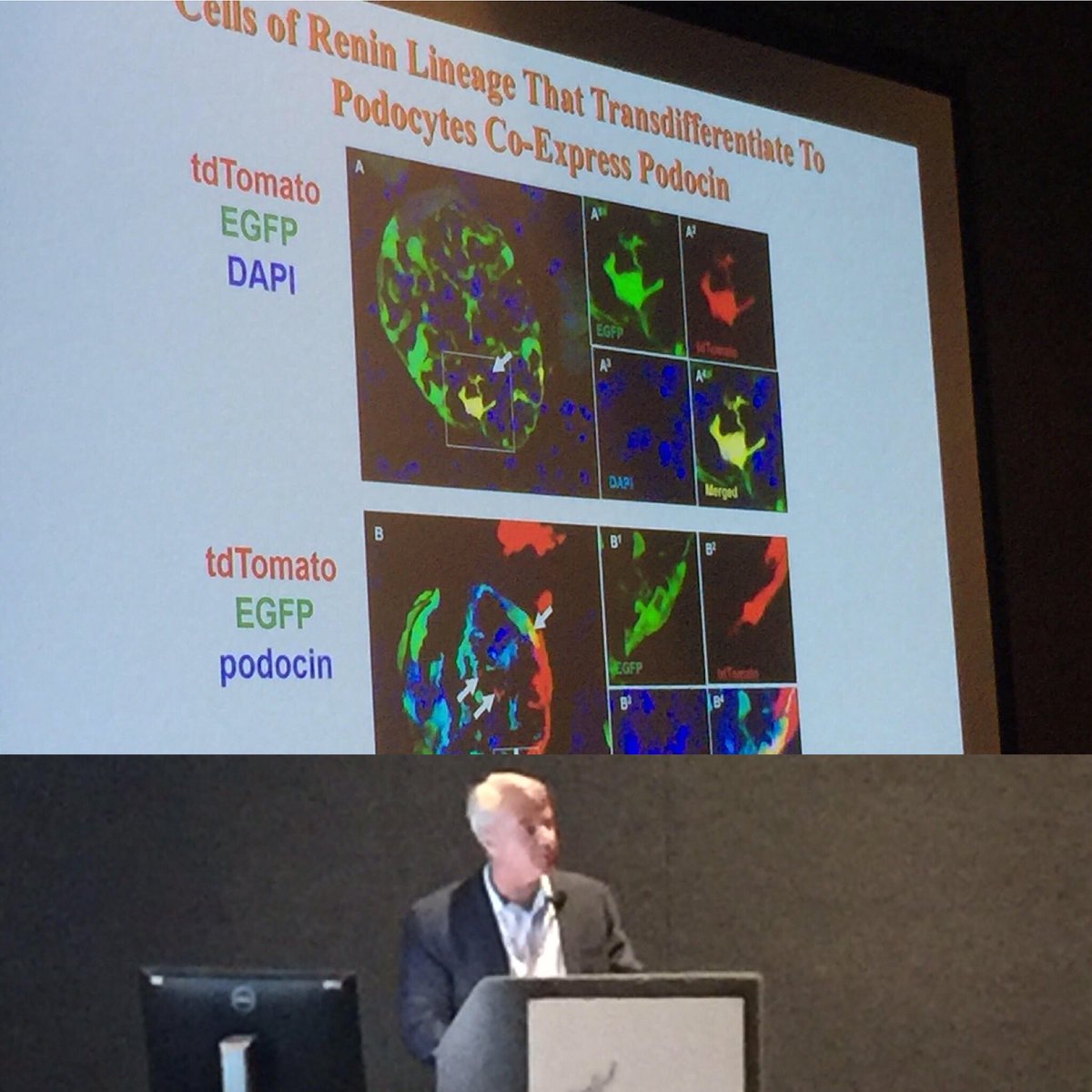
Kaverina, Natalya
Multi-Clonal Population of Cells of Renin Lineage (CoRL) Transdifferentiate into Podocytes and PECs in Experimental FSGS
Lichtnekert, Julia
RAS Inhibition Enhances Proliferation and Migration of Cells of Renin Lineage (CoRL) as Progenitors in Experimental FSGS